Nagi Bioscience recently won the CHF 10’000.- award at Philip Morris Equity Partner Startup Competition in Renens. So what does the leading tobacco company have in common with the start-up based at EPFL? The will to design a smoke free future by developing better alternatives to cigarettes for smokers, as we learned from the CEO of the start-up, Matteo Cornaglia.
End September, Nagi Bioscience, which is part of the MassChallenge accelerator program, won the Philip Morris Equity Partner (PMEP) Startup Competition in Renens. As Matteo Cornaglia explains, PMEP is the Corporate Venture Capital arm of Philip Morris International, one of the world leading tobacco companies. They invest in entrepreneurs and growth companies that could help contribute to PMI’s vision for a smoke free future.
Speaking of the competition organized at MassChallenge Switzerland, Nagi Biosicence CEO explains that it was divided into two steps: a first evaluation of the startup business case through a one-pager highlighting how our business could have an impact on PMI’s value chain and its smoke free future vision. Then, a pitching session with the 10 finalists was organized at PMI office in Lausanne with 10min pitch and 5min QA. Finally, a jury, headed by the PMI CSO Manuel C. Peitsch, evaluated the potential strategic fit with PMI & how the start-ups can impact their business.
Disruptive technology impresses jury
Nagi Bioscience’s project was selected because it developed a disruptive technology which will help companies, like PMI, to rapidly and ethically test their newly developed substances or chemicals. Indeed, each novel substance must be tested for their efficacy but also for their safety for humans and environment. This is particularly important in the PMI R&D process which must assess their smoke-free products to verify their potential to reduce individual health risk compared with continued smoking. In this context, in vivo animal testing, which is the current gold standard for this safety evaluation, is increasingly resource-consuming and ethical-issue-prone, more and more controlled by laws, often driven by public pressure. The introduction of their technology based on C. elegans as a valid alternative model in the PMI R&D process is meant to significantly reduce the number of animals used by this industry. Animal testing could be hence strongly limited and toxicity assays could be anticipated and reiterated along in the substance testing pipeline, to rapidly get precious in vivo toxicology information about the product under development.
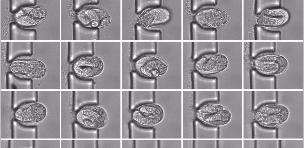
The 1st all-in-one “organism-on-chip” technology
Nagi Bioscience proposes the first all-in-one “organism-on-chip” technology for safety and efficacy testing of substances generated by pharma, cosmetic and chemical industries. The solution of the start-up includes a laboratory device which allows performing compounds testing on the micro-organism C. elegans, within disposable microfluidic chips. Their technology is meant to enter the rapidly growing industry of the emerging alternatives to animal testing, enabling for the first time “in vivo testing at the in vitro scale”. This concept directly defines the USP of their technology, which perfectly fills the current gap between cell-based and mice-based tests.
Speeding up development toward industrialization
Speaking of its future plans, the start-up run by its 3 co-founders recently hired a software engineer as well as an industrial designer in order to speed-up the technology development toward the future industrialization of its product. Based at the Ecole Polytechnique Fédérale de Lausanne (EPFL), Nagi Bioscience is currently looking for CHF 1.5M investment in order to accelerate the product development and pursue its pilot tests with several companies already interested by its technology. The goal is to reach the market by 2021.
(ES)























































Please login or sign up to comment.
Commenting guidelines